Frontal and motor cortex oxygenation during maximal exercise in normoxia and hypoxia
Leave a CommentAbstract
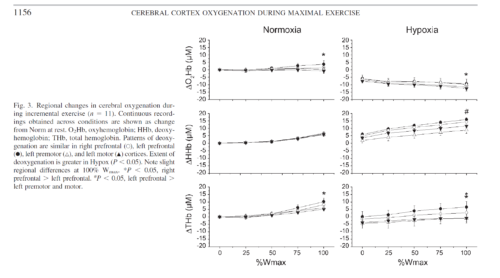
Reductions in prefrontal oxygenation near maximal exertion may limit exercise performance by impairing executive functions that influence the decision to stop exercising; however, whether deoxygenation also occurs in motor regions that more directly affect central motor drive is unknown. Multichannel near-infrared spectroscopy was used to compare changes in prefrontal, premotor, and motor cortices during exhaustive exercise. Twenty-three subjects performed two sequential, incremental cycle tests (25 W/min ramp) during acute hypoxia [79 Torr inspired Po(2) (Pi(O(2)))] and normoxia (117 Torr Pi(O(2))) in an environmental chamber. Test order was balanced, and subjects were blinded to chamber pressure. In normoxia, bilateral prefrontal oxygenation was maintained during low- and moderate-intensity exercise but dropped 9.0 +/- 10.7% (mean +/- SD, P < 0.05) before exhaustion (maximal power = 305 +/- 52 W). The pattern and magnitude of deoxygenation were similar in prefrontal, premotor, and motor regions (R(2) > 0.94). In hypoxia, prefrontal oxygenation was reduced 11.1 +/- 14.3% at rest (P < 0.01) and fell another 26.5 +/- 19.5% (P < 0.01) at exhaustion (maximal power = 256 +/- 38 W, P < 0.01). Correlations between regions were high (R(2) > 0.61), but deoxygenation was greater in prefrontal than premotor and motor regions (P < 0.05). Prefrontal, premotor, and motor cortex deoxygenation during high-intensity exercise may contribute to an integrative decision to stop exercise. The accelerated rate of cortical deoxygenation in hypoxia may hasten this effect.
Methods
Subjects.
After approval from the Colorado Multiple Institutional Review Board, 25 active, healthy volunteers (23 men and 2 women) from the Denver, CO, metropolitan area (elevation 1,650 m) provided written, informed consent to participate in a larger study investigating the etiology of acute mountain sickness. Physical examinations, including blood and urine tests, were conducted to verify general health before participation. All study procedures followed ethical guidelines established by the Declaration of Helsinki.
Study design.
Two incremental exercise tests were performed in an environmental chamber under ambient normobaric [normoxic (Norm), 610 Torr barometric pressure (Pb), 118 Torr inspired Po2 (PiO2)] and hypobaric [hypoxic (Hypox), 425 Torr Pb, 79 Torr PiO2] conditions to assess aerobic fitness for the larger study. Both tests were performed during a single chamber session to allow direct comparisons between Norm and Hypox without introduction of error from sensor placement/replacement. Tests were counterbalanced to control for order using a blinding strategy that varied chamber pressure to elicit similar sounds and changes in ear pressure during standardized 15-min ascent and descent periods. After arrival at the target Pb, 15 min were needed to adjust the cycle ergometer (Velotron Dynafit Pro, Racermate, Seattle, WA) and equip subjects with instrumental sensors (see below). Resting data were collected for 2 min before a 5-min warm-up at 50 W. Work rate was then incrementally increased using a 25 W/min ramp protocol to exhaustion. Subjects were blinded to elapsed time, power output, pedal revolutions per minute, and all physiological signals. Cool-down exercise was performed at 50 W for 5 min before chamber pressure was adjusted. After 15 min of ascent/decent and 15 min of rest at the second Pb, the protocol was repeated.
Results
Subjects.
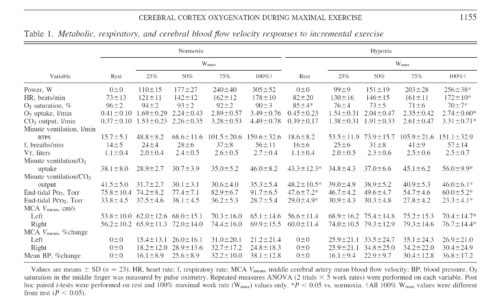
Twenty-three subjects (29 ± 8 yr of age, 73.7 ± 10.0 kg body wt, 181.7 ± 8.0 cm) completed both trials. Two subjects (with NIRS configuration 1) completed the Norm trial but were excluded from further study because of nausea and/or paresthesia during the subsequent depressurization period. Metabolic and power responses to both conditions were representative of physically fit, age-matched individuals (Table 1). Hypox reduced maximal V̇o2 and Wmax by 16 ± 6% and 21 ± 12%, respectively (P < 0.01). Order of trials did not affect the difference in maximal V̇o2 (P = 0.20) or Wmax (P = 0.95) between conditions, and 8 of 23 subjects were unable to retrospectively identify the order of testing.
Conclusions.
Cortical deoxygenation during high-intensity exercise is not restricted to prefrontal regions of the brain. Deoxygenation in premotor and motor cortices may contribute to fatigue and/or decisions to stop exercising. Acute hypoxia exacerbates cortical deoxygenation and, thus, may hasten these effects. Future functional NIRS studies are needed to expand our understanding of the role of cerebral activity in exhaustive whole body exercise.
Authors: Andrew W. Subudhi, Brittany R. Miramon, Matthew E. Granger, and Robert C. Roach
University of Colorado Altitude Research Center, Denver and Colorado Springs Campuses, Colorado Springs
published in: Journal of Applied Physiology 106: 1153–1158, 2009
Frontal and motor cortex oxygenation during maximal exercise in normoxia and hypoxia (pdf)