A Device for the Quantification of Oxygen Consumption and Caloric Expenditure in the Neonatal Range
BACKGROUND:
The accurate measurement of oxygen consumption (VO2) and energy expenditure (EE) may be helpful to optimize the treatment of critically ill patients. However, current techniques are limited in their ability to accurately quantify these end points in infants due to a low VO2, low tidal volume, and rapid respiratory rate. This study describes and validates a new device intended to perform in this size range.
METHODS:
We created a customized device that quantifies inspiratory volume using a pneumotachometer and concentrations of oxygen and carbon dioxide gas in the inspiratory and expiratory limbs. We created a customized algorithm to achieve precise time alignment of these measures, incorporating bias flow and compliance factors. The device was validated in 3 ways. First, we infused a certified gas mixture (50% oxygen/50% carbon dioxide) into an artificial lung circuit, comparing measured with simulated VO2 and carbon dioxide production (VCO2) within a matrix of varying tidal volume (4–20 mL), respiratory rate (20–80 bpm), and fraction of inspired oxygen (0.21–0.8). Second, VO2, VCO2, and EE were measured in Sprague Dawley rats under mechanical ventilation and were compared to simultaneous Douglas bag collections. Third, the device was studied on n = 14 intubated, spontaneously breathing neonates and infants, comparing measured values to Douglas measurements. In all cases, we assessed for difference between the device and reference standard by linear regression and Bland–Altman analysis.

RESULTS:
In vitro, the mean ± standard deviation difference between the measured and reference standard VO2 was +0.04 ± 1.10 (95% limits of agreement, −2.11 to +2.20) mL/min and VCO2 was +0.26 ± 0.31 (−0.36 to +0.89) mL/min; differences were similar at each respiratory rate and tidal volume measured, but higher at fraction of inspired oxygen of 0.8 than at 0.7 or lower. In rodents, the mean difference was −0.20 ± 0.55 (−1.28 to +0.89) mL/min for VO2, +0.16 ± 0.25 (−0.32 to +0.65) mL/min for VCO2, and −0.84 ± 3.29 (−7.30 to +5.61) kcal/d for EE. In infants, the mean VO2 was 9.0 ± 2.5 mL/kg/min by Douglas method and was accurately measured by the device (bias, +0.22 ± 0.87 [−1.49 to +1.93] mL/kg/min). The average VCO2 was 8.1 ± 2.3 mL/kg/min, and the device exhibited a bias of +0.33 ± 0.82 (−1.27 to +1.94) mL/kg/min. Mean bias was +2.56% ± 11.60% of the reading for VO2 and +4.25% ± 11.20% of the reading for VCO2; among 56 replicates, 6 measurements fell outside of the 20% error range, and no patient had >1 of 4 replicates with a >20% error in either VO2 or VCO2.
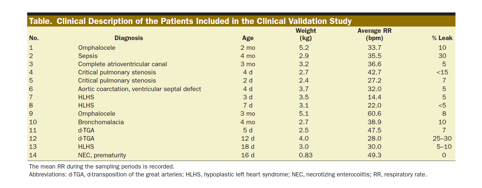
CONCLUSIONS:
This device can measure VO2, VCO2, and EE with sufficient accuracy for clinical decision-making within the neonatal and pediatric size range, including in the setting of tachypnea or hyperoxia.
Authors: Nachman, Einav BS*; Clemensen, Peter MS*,†; Santos, Katheryn BS*; Cole, Alexis R. BS*; Polizzotti, Brian D. PhD*,‡; Hofmann, Grace RRT§; Leeman, Kristen T. MD‡,∥; van den Bosch, Sarah J. MS*; Kheir, John N. MD*,‡
*Department of Cardiology, Boston Children’s Hospital, Boston, Massachusetts
†Department of Research and Development, InnoCC, Glamsbjerg, Denmark
‡Department of Pediatrics, Harvard Medical School, Boston, Massachusetts
Departments of §Respiratory Care
∥Newborn Medicine, Boston Children’s Hospital, Boston, Massachusetts



